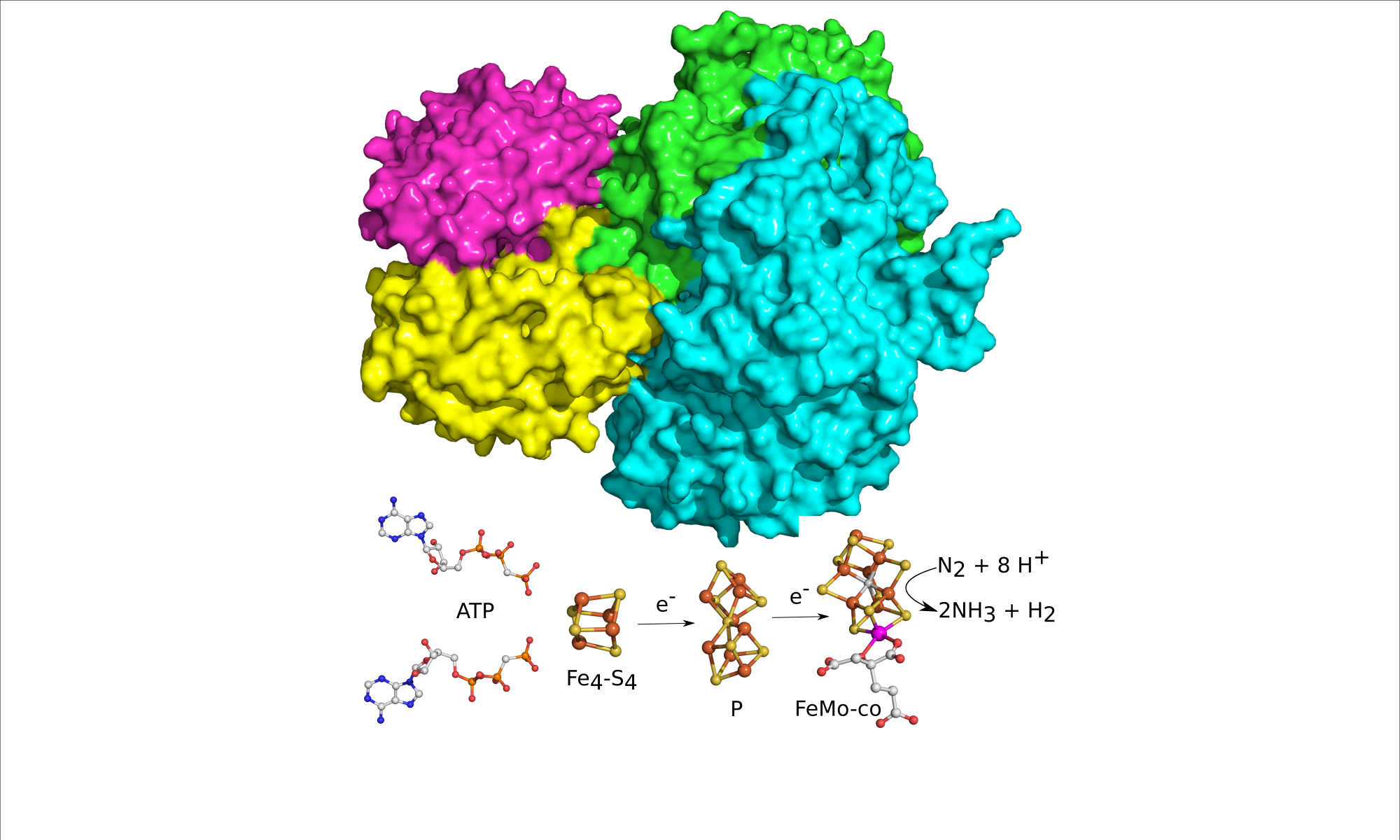
We are working to reveal the mechanism of the bacterial enzyme nitrogenase. This enzyme is responsible for the largest input of fixed N into the global biogeochemical N cycle. Nitrogenase carries out the chemically challenging reduction of N2 to NH3 under benign conditions of room temperature and atmospheric pressure. Our efforts are aimed at understanding how N2 is bound and reduced and how electrons are delivered in a reaction coupled to the hydrolysis of ATP.
Latest News
![]()
2016 “Negative cooperativity in the nitrogenase Fe protein cycle” published in Proc. Natl. Acad. Sci. USA. Article, News, USU Today
![]()
2016 Light-driven CO2 reduction in a photosynthetic bacterium catalyzed by nitrogenase published in Proc. Natl. Acad. Sci. USA. Article, News, News
![]()
2016 Research on light-driven N2 reduction by a CdS:MoFe protein hybrid published in Science. Article, News, News, Radio
2015 DOE funding, Biological Electron Transfer and Catalysis (BETCy), Energy Frontiers Research Center (EFRC), Department of Energy (DOE). News